Healthcare and life-science organizations — including medical technology providers, pharma manufacturers, clinical R&D teams, and medical device engineers — rely on Jira to coordinate sensitive and highly regulated projects. Typical workflows include:
-
Medical device development — design changes, validation, engineering updates.
-
Pharmaceutical R&D — tracking experiment results, approvals, and documentation.
-
Clinical trials — protocol updates, investigator queries, monitoring reports.
-
Quality & Compliance — non-conformance reports, audits.
In all these processes, every change must be clearly visible and fully documented. Transparent history is mandatory for compliance with standards such as 21 CFR Part 11, GxP, ISO 13485, and GDPR.
🎯 Goals
Healthcare organizations using Jira need to:
-
Maintain complete audit trails for every regulated workflow.
-
Provide traceable, exportable records for auditors and QA teams.
-
Track exactly who made which update and when.
-
Reduce manual effort during compliance checks and inspections.
-
Ensure data integrity and accountability across the lifecycle of devices, drugs, or patient records.
⚠️ Problems
Jira’s built-in history often fails healthcare audit requirements:
-
Changes can’t be exported in a way that's suitable for audits.
-
Deleted data (comments, description changes) is lost permanently.
-
Auditors must manually check multiple work items.
-
No unified view of field changes across regulated projects.
-
Difficult to demonstrate a consistent audit trail during inspections.
This leads to stressful audits, gaps in documentation, and increased compliance risks.
💡 Solution
Issue History for Jira app provides healthcare teams with the visibility and traceability required for regulatory compliance:
-
Shows complete chronological change history in a single table.
-
Tracks what changed, who changed it, and when across any project.
-
Supports CSV, Excel, and PDF exports for audits.
-
Allows filtering by project, user, time period, or specific fields.
-
Records deleted work items and restores them in seconds, giving visibility that Jira doesn’t.
Many mid-size and enterprise healthcare companies already use Issue History to keep device engineering, medical software, pharma R&D, and clinical operations fully compliant and inspection-ready.
🔄 Workflow Examples
1. Medical Device Design Controls
Use case: A medical device engineering team must prove that all design changes were reviewed, approved, and documented.
How Issue History for Jira helps:
-
Allows filtering Jira work items for status changes, description edits, deadline updates, and approval steps.
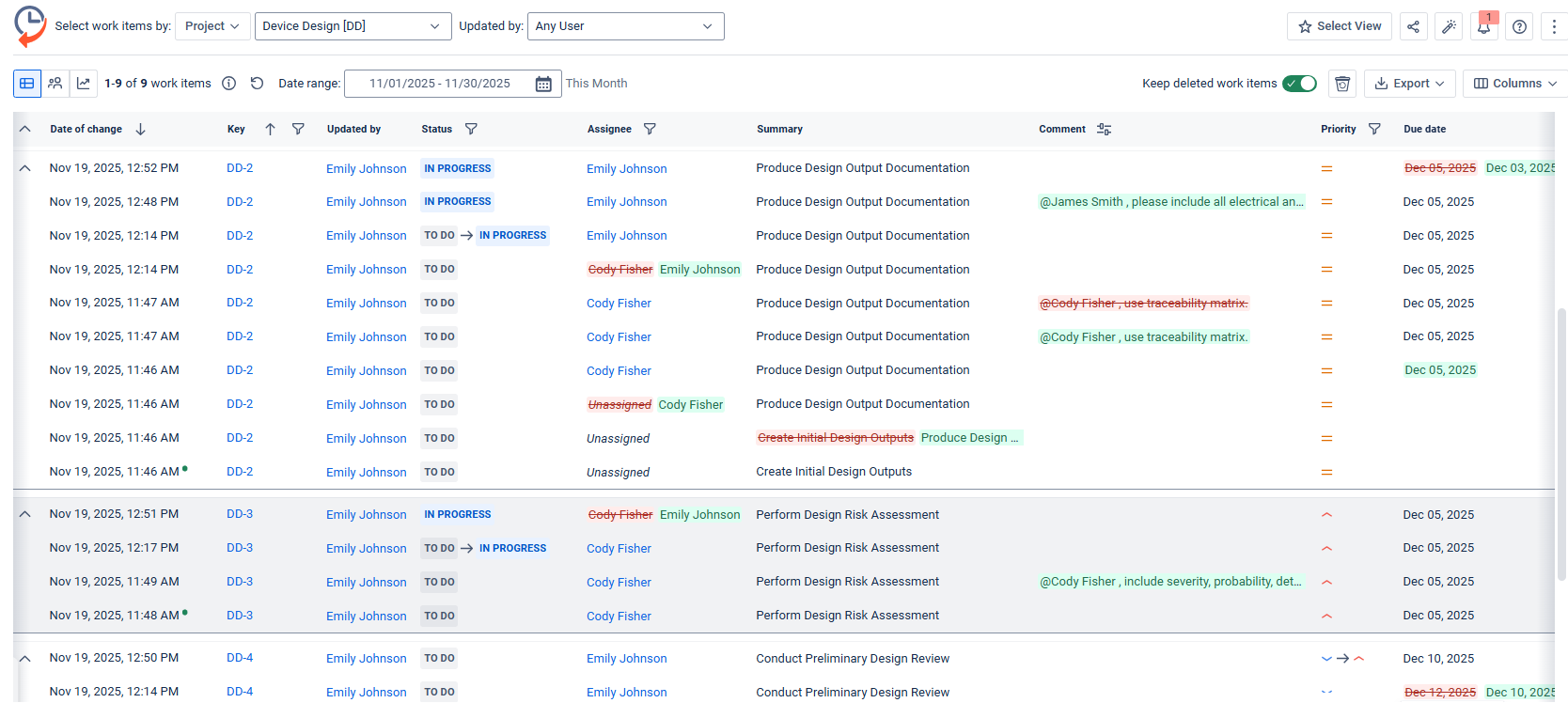
-
They export chronological change logs to attach to Design History Files (DHF).
.png?cb=e6b2625a243535ae20a4a32aa45136ce)
-
Inspectors can see exactly how the design evolved and who was responsible for each step.
.png?cb=3bd9ef75d4210564d4e0de0394bafb82)
2. Pharmaceutical R&D and Lab Data Integrity
Use case: Scientists must ensure experimental data and SOP updates meet data-integrity standards.
How Issue History for Jira helps:
-
All edits to descriptions and fields are captured.
-
Deleted or overwritten comments remain visible in the change table.
-
Research leads to an export-ready, clean, audit-ready history.
With Issue History for Jira app, our clients in the healthcare sector can save time on audits, reduce risks, and ensure every Jira project is fully accountable.
Haven't used this app yet? 👉 Then you’re welcome to try it 🚀